The Versatile Substance That Is Carbon Black
I suspect very few of us think much about carbon black.
In an over fifty-year working life, I have only come across carbon black indirectly and no-one has actually shown me any carbon black.
This is the first sentence of the Wikipedia entry for carbon black.
Carbon black (with subtypes acetylene black, channel black, furnace black, lamp black and thermal black) is a material produced by the incomplete combustion of coal tar, vegetable matter, or petroleum products, including fuel oil, fluid catalytic cracking tar, and ethylene cracking in a limited supply of air.
It doesn’t sound the most appetising of substances and the next sentence reinforces that view.
Carbon black is a form of paracrystalline carbon that has a high surface-area-to-volume ratio, albeit lower than that of activated carbon. It is dissimilar to soot in its much higher surface-area-to-volume ratio and significantly lower (negligible and non-bioavailable) polycyclic aromatic hydrocarbon (PAH) content.
The text is illustrated with what looks like a small pile of soot.
I first came across carbon black, in my first job after leaving Liverpool University at ICI Mond Division at Runcorn.
For a time, I shared an office with Peter, who was part of a number of engineers, who were trying to get a new plant, that had been purchased from BASF to make commercial quantities of acetylene. All the plant seemed to make was large quantities of soot, which it then proceeded to spread all over the town of Runcorn.
If I remember correctly, the process worked by burning ethylene in a limited supply of air and then quenching it with naphtha. The similarities between the BASF process and the method for producing carbon black lead me to believe, that ICI’s process was probably producing a lot of carbon black.
Peter was working on an instrument that measured the quantity of acetylene in the off-gas from the burners and he succeeded, but unfortunately proved that the plant was going into explosive limits. For this reason, ICI shut their process, although BASF persevered.
Ethylene is a hydrocarbon which has the formula C2H4 or two carbon and four hydrogen atoms. So if you can get them to stop tightly holding hands with no oxygen around, the hydrogen will pair off as H2 and the carbon will exist as a lot of single C atoms or carbon black.
BASF and ICI were trying to produce acetylene or C2H2, where there is a powerful triple bond between the two carbon atoms. All that energy in the acetylene makes it useful for activities like welding.
Common Uses Of Carbon Black
The Wikipedia entry for carbon black, has this summary of its uses.
The most common use (70%) of carbon black is as a pigment and reinforcing phase in automobile tires. Carbon black also helps conduct heat away from the tread and belt area of the tire, reducing thermal damage and increasing tire life. Its low cost makes it a common addition to cathodes and anodes and is considered a safe replacement to lithium metal in lithium-ion batteries. About 20% of world production goes into belts, hoses, and other non-tire rubber goods. The remaining 10% use of carbon black comes from pigment in inks, coatings, and plastics, as well as being used as a conductive additive in lithium-ion batteries.
The entry then gives a list of other uses, some of which are still being developed.
Global Production Of Carbon Black
This paragraph is from the Wikipedia entry for carbon black.
Total production was around 8,100,000 metric tons (8,900,000 short tons) in 2006. Global consumption of carbon black, estimated at 13.2 million metric tons, valued at US$13.7 billion, in 2015, is expected to reach 13.9 million metric tons, valued at US$14.4 billion in 2016.
So we have the useful paradox, that we don’t want to emit more carbon dioxide, but extra carbon black could probably be usefully used.
Conclusion
Using the HiiROC process to extract hydrogen could even give us a biproduct ; carbon black, that has uses.
Hydrogen Energy Explained
The title of this post, is the same as this story on Centrica.
This is the sub-heading.
Hydrogen is a clean alternative to natural gas, when it’s burnt it doesn’t produce carbon dioxide, which is a harmful greenhouse gas. It has the potential to play a significant role in achieving net-zero and decarbonising various sectors.
The story is a good introduction to hydrogen.
In the 1960s, when I first worked in a hydrogen electrolyser at ICI in Runcorn, I would have found a document like this one from Centrica invaluable.
Observing Trains At Stafford Station
I’m fairly certain, that I’ve only been to Stafford once in my life, in the past. That would be in 1968, when I went to English Electric in the town, when I was looking for a job on leaving Liverpool University.
In the end, my first job on leaving was at ICI in Runcorn.
As Stafford station, is on the West Coast Main Line, I’ve been through the station many times, on my way to Liverpool, Manchester, Preston, Scotland and other places in the North West of England.
I travelled North to Stafford in one of London Northwestern’s Class 350 trains, which definitely had ironing board seats. Some of these trains are being replaced with new Class 730 trains.
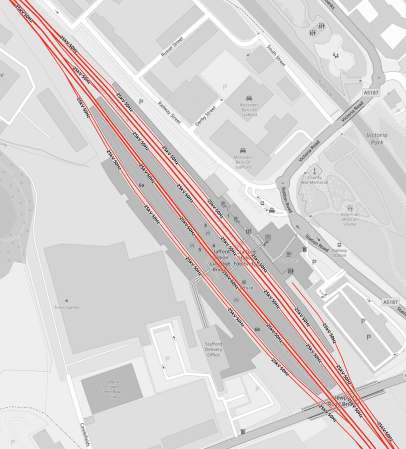
Stafford Station
This OpenRailwayMap shows the layout of platforms at the station.
Note.
- Platforms are numbered starting with Platform 1 in the East.
- There are five through tracks for passenger trains and a double-track goods loop.
- All tracks are electrified and bi-directional.
- The platforms are three hundred metres long, but I feel they could be lengthened to take the 400 metre long High Speed Two trains.
- The platforms are certainly long enough for pairs of 200 metre long High Speed Two trains, to split and join at the station.
These pictures show Stafford station.
Note.
- The station was built of concrete in the early 1960s.
- It has a unique feature, in that there is a waiting area above the tracks, so kids of all ages can watch the trains speed through.
- It has been improved several times in recent years.
- It is step-free with several lifts.
I feel, the station could be converted into a major High Speed Two hub station.
High Speed Two Services
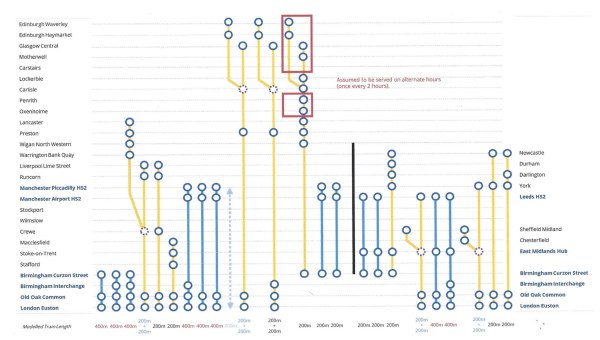
This graphic shows High Speed Two services after Phase 2b is completed.
Note.
- Everything to the right of the vertical black line has now been cancelled
- Services shown in blue are run by High Speed Two Full Size trains.
- Services shown in yellow are run by High Speed Two Classic-Compatible trains.
- Each of these trains will have a frequency of one train per hour (tph).
These High Speed Two trains pass through Stafford.
- London and Lancaster/Liverpool Lime Street – Split/Join at Crewe
- London and Crewe/Runcorn/Liverpool Lime Street
- London and Stafford/Stoke-on-Trent/Macclesfield
- London and Manchester Airport/Piccadilly
- London and Manchester Airport/Piccadilly
- London and Manchester Airport/Piccadilly
- London and Edinburgh/Glasgow – Split/Join at Carlisle
- London and Edinburgh/Glasgow – Split/Join at Carlisle
- Birmingham and Edinburgh/Glasgow
- Birmingham and Manchester Airport/Piccadilly
- Birmingham and Manchester Airport/Piccadilly
In addition space will need to be found for these other services.
- Avanti – London and Chester/North Wales
- Avanti – London and Blackpool
- Freight – London and Liverpool/Manchester and Scotland
That gives a total of at least fourteen tph through Stafford station or one every 257 seconds.
Between Stafford and Crewe, with the completion of the flyover at Norton Bridge, there is effectively four tracks all the way, so this should give enough capacity to allow fourteen tph between Stafford and Crewe.
Between Stafford and Handsacre junction, where High Speed Two will join the Trent Valley Line about half the route is four-track with the rest being just two-track.
Will the two-track section be able to handle fourteen tph? I suspect it will, especially, as it is sandwiched between two four-track sections.
These are my thoughts about making the most of capacity through Stafford station.
Could Pairs Of High Speed Two Trains, Split And Join At Stafford?
Stafford station has 300 metre long platforms and pairs of High Speed Two Trains are 400 metres long.
Looking at maps and pictures of Stafford station, I suspect that lengthening the platforms at Stafford would be possible.
In the provisional High Speed Two service diagram, that I displayed earlier, these are two services.
- London and Crewe/Runcorn/Liverpool Lime Street
- London and Stafford/Stoke-on-Trent/Macclesfield
These could be run as a pair of trains between London and Stafford.
One advantage of running the two services as a pair, is that it would reduce the number of trains between Stafford and Handsacre junction by one, which might help the scheduling of trains through the double-track section, of the Trent Valley Line.
Fitting In The London and Chester/North Wales Services
Consider.
- These important services will soon be run by five-car Class 805 trains.
- The trains are diesel-electric.
- A pair would be 260 m long.
- There are ten trains per day (tpd) on the route.
The service has a few problems.
- It takes up a path between Crewe and Handsacre junction via Stafford, where train paths are at a premium.
- It is not zero-carbon.
- If London and Holyhead times were reduced, it would probably attract more passengers to the route.
As there are plenty of spare paths on High Speed Two, due to the cancellation of the Eastern leg, the easiest way to speed up the service would be to use High Speed Two between Handsacre junction and London.
200 metre long High Speed Two Classic-Compatible trains could be used, but this would need one of the following.
Electrification between Crewe and Holyhead. Although this was promised by the last government, I suspect the antis will stop it.
Partial electrification between Crewe and Holyhead. As Crewe and Holyhead is only 105.5 miles, I suspect a creditable scheme to use battery-electric trains could be developed. These trains could be useful in opening up High Speed Two to other destinations.
Could Other Services Piggy-Back On London and Chester/North Wales Services?
The London and Chester/North Wales services would all be run by 200 metre long High Speed Two trains.
There would be no reason, why on the London end of the journey, they could be paired with another High Speed Two train to and from another destination.
- They could split and join with a Blackpool service at Crewe.
- They could split and join with a Barrow service at Crewe.
- They could split and join with a second Stoke-on-Trent/Macclesfield service at Stafford.
Services like these, would improve the coverage of High Speed Two.
As with the joining of the Liverpool Lime Street and Macclesfield services, the pairing of trains reduces the number of paths needed on the double-track section of the Trent Valley Line to the South of Stafford.
Other Splitting And Joining At Stafford
It may be possible to arrange other splitting and joining at Stafford.
By rearranging the Manchester and/or the Scottish services, it might be possible to reduce the number of trains, through the double-track section of the Trent Valley Line.
There are certainly several possibilities.
Conclusion
Stafford station could be crucial, in making High Speed Two Lite feasible.
Was I One Of The First To Have My Temperature Measured By A Thermometer Reading The Radiation From My Ear?
Last week, a doctor read my body temperature, by using an electronic thermometer, that read the temperature inside my ear.
But it wasn’t the first time!
That must have been in 1968 or 1969, when I was working at ICI in Runcorn.
ICI had a problem, in that they needed to read the temperature of chemical reaction vessels.
- Temperatures could be higher, than 1,000 °C.
- Some mixtures could be highly corrosive.
- Safety needed to be as high as possible.
My colleague; John Baxendale was assigned the problem.
John came up with a solution based on black bodies and their unique black body radiation.
These two paragraphs, from the Wikipedia entry for black body, explain the principle.
A black body or is an idealised physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence. The radiation emitted by a black body in thermal equilibrium with its environment is called black-body radiation. The name “black body” is given because it absorbs all colours of light. In contrast, a white body is one with a “rough surface that reflects all incident rays completely and uniformly in all directions.”
A black body in thermal equilibrium (that is, at a constant temperature) emits electromagnetic black-body radiation. The radiation is emitted according to Planck’s law, meaning that it has a spectrum that is determined by the temperature alone, not by the body’s shape or composition.
Note, that I have very mildly edited, what Wikipedia says, to the King’s English.
John had developed some clever electronics, that read the spectrum of the radiation and by decoding the spectrum, he was able to calculate the temperature.
Early on in the testing, John found that nearly all of us, have two black bodies on the side of our heads; our ears, so he could measure the temperature inside them.
The Aerosol Tales
When I left Liverpool University in 1968, I was very familiar with the use of products distributed in aerosol cans.
- I had used aerosol shaving cream, although about that time, I acquired my beard.
- I certainly used aerosol deodorant, as did most in the 1960s.
- Aerosol paints were common for covering scuffs and scratches in your car.
- Aerosols were often used to apply sun protection.
- Aerosols containing cream or a non-dairy alternative for culinary use were not unknown.
- Aweosol lubricants were starting to appear.
Although, I went to work for the chemical giant; ICI, at that time, I had no idea how an aerosol and its can worked.
As ICI at the time, ICI were major manufacturers of aerosol propellants, I quickly learned how they worked.
The Wikipedia entry for Aerosol Spray Dispenser gives a lot of history about aerosol cans and their propellants.
The Wikipedia entry for Propellant has this paragraph describing propellants of the last century.
Chlorofluorocarbons (CFCs) were once often used as propellants, but since the Montreal Protocol came into force in 1989, they have been replaced in nearly every country due to the negative effects CFCs have on Earth’s ozone layer. The most common replacements of CFCs are mixtures of volatile hydrocarbons, typically propane, n-butane and isobutane. Dimethyl ether (DME) and methyl ethyl ether are also used. All these have the disadvantage of being flammable. Nitrous oxide and carbon dioxide are also used as propellants to deliver foodstuffs (for example, whipped cream and cooking spray). Medicinal aerosols such as asthma inhalers use hydrofluoroalkanes (HFA): either HFA 134a (1,1,1,2,-tetrafluoroethane) or HFA 227 (1,1,1,2,3,3,3-heptafluoropropane) or combinations of the two. More recently, liquid hydrofluoroolefin (HFO) propellants have become more widely adopted in aerosol systems due to their relatively low vapor pressure, low global warming potential (GWP), and nonflammability.
Note that the whole range of these chemicals, effect the ozone layer.
Rocksavage Works
ICI’s Rocksavage Works, was an integrated chemical plant by the Mersey,.
- It made all types of CFCs for aerosols and other purposes.
- It also made the fire suppressant and extinguisher; Bromochlorodifluoromethane or BCF.
- Alongside BCF, it made the anaesthetic Halothane or as ICI called it Fluothane.
- The plant was a poisonous place with all those bromine, chlorine and fluorine compounds.
- Despite this, the plant had a remarkable safety record.
I had the pleasure of working at the plant and it was where, I had most of my excellent Health and Safety training, from the amazing site foreman; Charlie Akers.
Some of the wisdom he distributed has proved invaluable in aiding my stroke recovery.
I suspect that since the signing of the Montreal Protocol, the plant has changed greatly or has even been closed.
All that appears to be left is the 800 MW gas-fired Rocksavage power station and a Facebook page.
Aerosol Baked Beans
In those days, I worked most of the time in a lab at Runcorn Heath.
One of the labs near to where I generally worked, in the large research complex, was a lab, where new aerosol products were developed and tested.
One of the standard jokes about that lab, was that they were working on aerosol baked beans. They said, they would develop the product, even of they had to eject them from the can one at a time.
Gift Time
One afternoon, the boss of the aerosol development lab came through with a tray of goodies.
On the tray, which was much like a cinema usherette’s ice cream tray of the sixties was a whole host of partly-labeled aerosol cans. Only clues to what the product might be were written on the outside in felt-tip pen.
I grabbed two, one of which was marked something like lubricating oil and the other was just marked hand cream, which I of course gave to my new wife; C.
We were married for nearly forty years and often, when she bought hand cream, she would remark, that it wasn’t of the same standard as the little can I brought home from work.
It appears to me, that one of the world’s top cosmetic companies and ICI were trying to create the world’s best and probably most expensive hand creams.
DMW
Fast-forward nearly twenty years and I was approached by Lloyds Bank about two individuals, who had developed an aerosol valve, that instead of using CFCs or other ozone-depleting chemicals.
- By the exploitation of the nether end of fluid dynamics, the propellant of the aerosol was nothing more harmless than pure nitrogen.
- I formed a company called DMW with the two inventors.
- John Gummer, who at the time was my MP and Environment Minister, knew of the aerosol valve and he took the details to Montreal.
So did a device developed in Suffolk help push through the Montreal Protocol?
Osbourne Reynolds
I also wonder, if we had some supernatural help. At the time, I lived in the family home of Osbourne Reynolds.
- He did a lot of the early work on fluid dynamics.
- He was the first UK Professor of Engineering.
- He was professor of Engineering at Manchester University for nearly forty years.
- The Reynolds number is named after him.
- Remarkably, students are sill taught on the equipment Reynolds designed.
- Reynolds was certainly one of our great Victorian scientists.
This Wikipedia entry gives more details of his remarkable life and work.
After Montreal the aerosol valve was sold to Johnson & Johnson.
DMW continued to develop other products and we had one, who no-one had any idea about how it worked.
So I discussed it with the Reynolds’s expert at Manchester University and he said he had no idea either.
But he was absolutely certain, that Reynolds would have known.
Are Bodies Cleverer Than We Think?
Because, I had a serious stroke thirteen years ago, I am on Warfarin for life to thin my blood, so that I don’t have another stroke.
INR is short for International Normalized Ratio, which diverts on Wikipedia to the Wikipedia entry for Prothrombin Time, where these is the first two paragraphs.
The prothrombin time (PT) – along with its derived measures of prothrombin ratio (PR) and international normalized ratio (INR) – is an assay for evaluating the extrinsic pathway and common pathway of coagulation. This blood test is also called protime INR and PT/INR. They are used to determine the clotting tendency of blood, in such things as the measure of warfarin dosage, liver damage, and vitamin K status. PT measures the following coagulation factors: I (fibrinogen), II (prothrombin), V (proaccelerin), VII (proconvertin), and X (Stuart–Prower factor).
PT is often used in conjunction with the activated partial thromboplastin time (aPTT) which measures the intrinsic pathway and common pathway of coagulation.
That is very technical and complicated, but what does it mean practically?
Those on Warfarin to thin their blood are generally supposed to keep their INR between 2 and 3.
I test the INR myself with a Coaguchek meter from Roche and have done so for nearly ten years.
Every, so often, I discuss the level of Warfarin with my GP and we adjust the level accordingly.
Typically, NHS patients on Warfarin have their INR checked every few weeks.
But as I am a Graduate Control Engineer, who has written control strategies for chemical plants, I feel this checking every few weeks, is not enough.
At times, I have checked at a higher frequency.
Four times, I have had minor operations.With the first operation, I agreed with the surgeon, that I would lower my INR to 2.1 for the operation and raise it back to 2.5 after the operation.
It all went well and I repeated the exercise for the three other minor operations.
I have checked daily since about the beginning of April, before which, I generally tested on Mondays and Thursdays.
There were two reasons for the change of frequency.
- Regular storms seemed to roll in and for some reason, they tended to lower my INR.
- I also wrote Do Thunderstorms Cause Strokes?, after reading a paper from the United States about the relationship between thunderstorms and strokes.
So I took the prudent decision to test my INR daily.
Then on the 30th May, disaster struck.
My Coaguchek meter found out it had the wrong firmware and Roche implored me to change it.
But I am not a hardware person, so I was left unable to check my INR.
Luckily, I remembered a story from the 1960s about one of ICI’s chemical plants.
This plant was one of the first fully-digitally controlled plants controlled by an IBM 1800 computer.
It also ran 24 hours a day, seven days week.
So in the middle of the night, the plant operators got out all the computer printouts, which showed how the computer had set all the valves and controllers.
One-by-one they reset all the controls on the plant to the settings that the computer had used for the last few days.
Using, this computer-assisted mode the plant was kept running, until engineers could fix the computer.
On the 30th May, my INR was a little bit high at 2.9 and I was using a dosage of Warfarin of 4 mg. one day and 3.5 mg. the next.
The strange dosage was one, that I know from experiment over time produces an INR of 2.5.
My actual average Warfarin dose was 3.73 over the last thirty days and my INR, as measured most days was 2.6 over the same period.
So, I did what the engineers did on the chemical plant, kept calm and carried on.
Yesterday my son fixed the Coaguchek meter and I was able to take my first reading for 17 days. It was 2.7 and only 0.1 higher than the 30-day average on the 30th May.
I had successfully jumped the gap in the readings.
Conclusion
Most systems have an equilibrium. Make sure you know it.
The Chemical Engineer Magazine On Highview Power
This is said in the Wikipedia entry for The Chemical Engineer Magazine.
The Chemical Engineer is a monthly chemical engineering technical and news magazine published by the Institution of Chemical Engineers (IChemE).
I first read the magazine perhaps fifty years ago, when it explained a chemical process, I was working on at ICI.
It is one of several well-respected magazines published by UK scientific, medical and engineering institutions.
This article in the magazine is entitled Energy Stored As Liquefied Air: £300m Investment Triggers Construction Of UK’s First Commercial-Scale Plant.
These three paragraphs introduce the investment.
HIGHVIEW POWER has received £300m (US$379m) in funding to build the UK’s first commercial-scale liquid air energy storage plant (LAES), designed to balance peaks and troughs in power demand as more renewable energy sources are brought online.
Construction of the facility is now underway in Carrington near Manchester. It is scheduled to begin operations in early 2026 and the company predicts it will provide more than 700 jobs in construction and the supply chain.
The plant will have a storage capacity of 300 MWh and an output of 50 MW per hour for six hours.
The rest of the article is just two sections.
- How Will Highview’s New Plant Work?
- Why Is The Technology Needed?
This article in the magazine is an absolute must read.
Bilfinger Drives Highview Power’s Innovative Storage Project, Accelerating The Energy Transition
The title of this post, is the same as that of this press release from Bilfinger, who are a European multinational engineering and services company.
This is the first paragraph of the Wikipedia entry for Bilfinger.
Bilfinger SE (previously named Bilfinger Berger AG) is a European multinational company specialized in civil and industrial construction, engineering and services based in Mannheim, Germany.
Fifty years ago, I was playing a very small part in the designing of complex chemical plants for ICI. My part was mainly to check, the mathematics and dynamics for the designs, the engineers wanted to use.
The experience certainly left me with the belief, that to design a world-class chemical plant is not an easy process.
So if I was needing a complex chemical plant, I would call in the experts.
A Highview Power energy storage system, may not be a chemical plant, but it shares many of the factors of chemical plants, waterworks and sewage plants. So building one, needs a company, with wide experience, which Bilfinger certainly appear to have.
This paragraph from the press release, summarises Bilfinger’s roll in Hghview Power’s Manchester project to create a 50 MW/300 MWh battery based on Highview’s proprietary long duration energy storage system.
The scope of Bilfinger’s services ranges from the procurement of steel to extensive plant construction services, including mechanical, electrical, instrumentation, insulation, painting and structural steel work, along with the overall management of all aspects of construction, including civil works and equipment installation. As principal contractor, Bilfinger is committed to ensuring the highest safety standards on site.
In UK Infrastructure Bank, Centrica & Partners Invest £300M in Highview Power Clean Energy Storage Programme To Boost UK’s Energy Security, I described how Highview Power had recruited high-class backers to fix the companies finances.
It looks like the Bilfinger deal to build the first system, is the last piece of the jigsaw and will see Highview Power on its way.
The Problem Of Waste Plastic And Why Pyrolysis Oil Might Just Contain The Answer
The title of this post, is the same as that of this article on the Chemical Engineer.
These three paragraphs introduce the article.
One of the few technologies that can break down unrecyclable post-consumer waste plastic, pyrolysis is fast becoming a potential recycling route for companies trying to reduce their waste output.
The world produces around 450m t/y of plastic, but only 9% is recycled, with most waste ending up in landfill. Pyrolysis, which involves heating the plastic at extremely high temperatures in the absence of oxygen, breaks down the molecules to produce pyrolysis oil or gas. The oil can then be used to develop new products.
George Huber, a professor of chemical engineering at the University of Wisconsin-Madison, is leading a research team that is investigating the chemistry of pyrolysis oil and its use in polyolefin recycling.
This is a quote from George Huber
Waste plastic should be viewed as a resource we can use to make plastics and other chemicals. We should not be landfilling or burning it, we should be reusing the carbon in waste plastics.
I very much agree with what he said.
These are my thoughts.
Pyrolysis
The Wikipedia entry for pyrolysis starts with this paragraph.
The pyrolysis (or devolatilization) process is the thermal decomposition of materials at elevated temperatures, often in an inert atmosphere.
This paragraph describes the technique’s use in the chemical industry.
The process is used heavily in the chemical industry, for example, to produce ethylene, many forms of carbon, and other chemicals from petroleum, coal, and even wood, or to produce coke from coal. It is used also in the conversion of natural gas (primarily methane) into hydrogen gas and solid carbon char, recently introduced on an industrial scale. Aspirational applications of pyrolysis would convert biomass into syngas and biochar, waste plastics back into usable oil, or waste into safely disposable substances.
I came across pyrolysis in my first job after graduating, when I worked at ICI Runcorn.
ICI were trying to make acetylene in a process plant they had bought from BASF. Ethylene was burned in an atmosphere, that didn’t have much oxygen and then quenched in naphtha. This should have produced acetylene , but all it produced was tonnes of black soot, that it spread all over Runcorn.
I shared an office with a guy, who was using a purpose-built instrument to measure acetylene in the off-gas from the burners.
When he discovered that the gas could be in explosive limits, ICI shut the plant down. The Germans didn’t believe this and said, that anyway it was impossible to do the measurement.
ICI gave up on the process and demolished their plant, but sadly the German plant blew up.
I would assume we have progressed with pyrolysis in the intervening fifty years.
University of Wisconsin-Madison
The University of Wisconsin-Madison is a top-ranked American University and is part of my daily life, as the Warfarin, that stops me having another stroke was developed at the University in the 1940s.
Conclusion
The article is a must-read and I feel that my past experience says, that George Huber and his team could be on to something.
I wish them the best of luck.